Latest Group News
Congratulations to Dr. Nathan Eden |
|
Porous polymers PTMSP and particle PAF-1 provides papers and patent
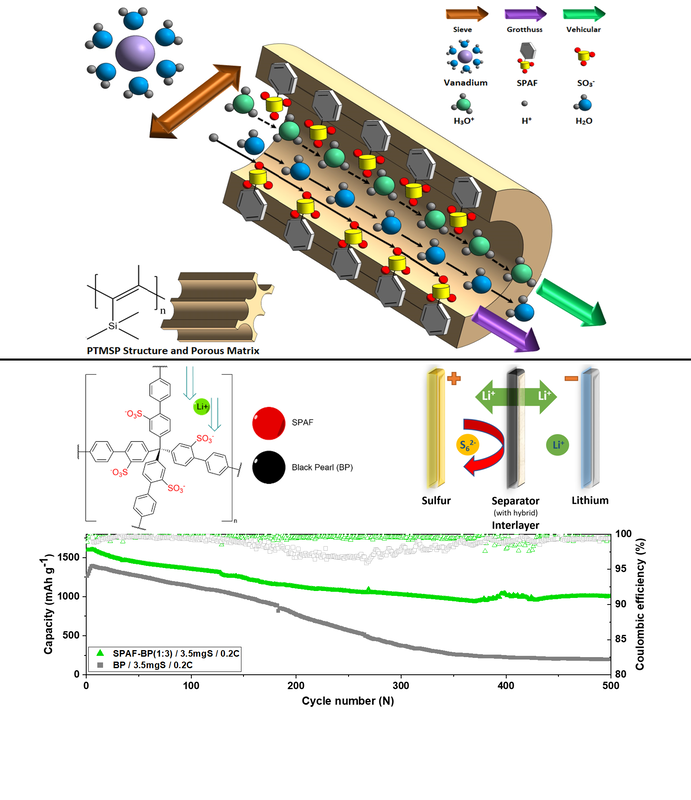
Congratulations to Ehsan publishing two papers on polymer membranes, used in lithium-sulfur batteries and vanadium flow batteries. A patent relating to this work was also filed. These results are described in the Journal of Materials Chemistry A Paper "Exceptional lithium diffusion through porous aromatic framework (PAF) interlayers delivers high capacity and long-life lithium–sulfur batteries" and ACS Applied Energy Materials Paper "Charge Carrier Molecular Sieve (CCMS) Membranes with Anti-aging Effect for Long-Life Vanadium Redox Flow Batteries" Vanadium crossover hinders widespread adoption of Vanadium Redox Flow Batteries (VRFBs). Superglassy polymer separators could control crossover but lack the requisite proton conductivity and cycling stability. We report a PTMSP/PAF-1-SO3H nanocomposite membrane that hinders crossover, with ideal PAF-1-SO3H pores delivering conductivity and aging suppression. |
|
Glucose in a battery? Sweet!
Congratulations to Yingyi on the publication of her Nature Communications paper "A saccharide-based binder for efficient polysulfide regulations in Li-S batteries". Yingyi's work developed a facile technique for improving the capacity retention and sulfur utilization in carbon-sulfur cathodes within lithium sulfur batteries. Her work has been reported on by the ABC. |
|
Highest carbon capture and storage efficiency recorded with new magnetic sponges
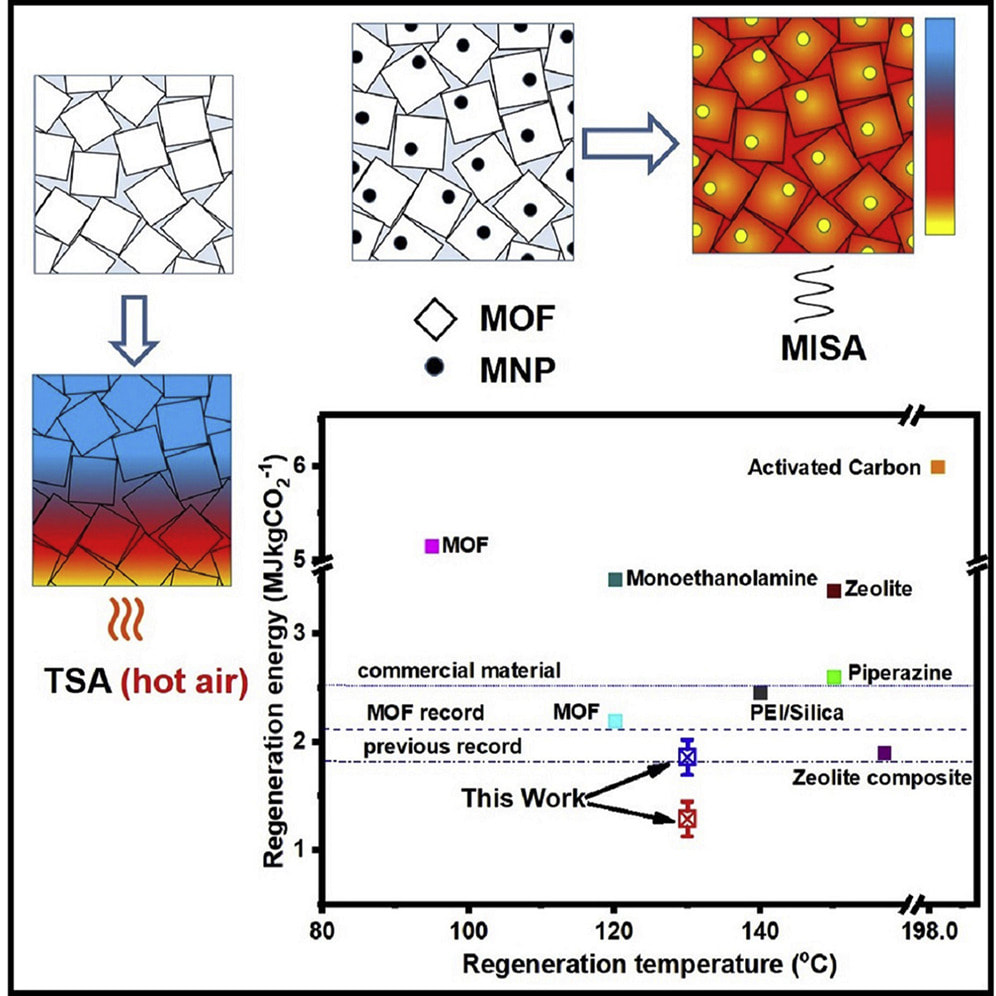
A/Prof Matthew Hill and Dr Muhammad Munir Sadiq have led a research effort to develop new advanced materials to capture CO2 from the air. M-74 CPT@PTMSP is able to both capture CO2 and efficiently release it due to the incorporation of magnets that allow for induction heating. This material would be able to be incorporated into a device to remove CO2 from the air as a negative emissions technology, and provide it as a feed stream into other processes, or stored. Importantly, it holds the record for lowest regeneration energy required per mass of CO2 for all advanced porous materials. |
|
Supercharged lithium-sulfur batteries ramping from lab to industry
Dr Mahdokht Shaibani, along with Prof Mainak Majumder and A/Prof Matthew Hill and their research team have developed an ultra-high capacity lithium-sulfur battery that has higher performance and lower environmental impact. The new batteries have enough charge to power a smartphone for 5 days continuously. As well as providing fundamental science, cells are being fabricated in the Fraunhofer Institute for Material and Beam Technology for larger scale testing and production runs. |
|
'Magic Crystals' developed in Australia to provide a boost to lithium production
A/Prof Matthew Hill received the Solomon Award for developing 'magic crystals' (Metal-Organic Frameworks) for the use of separation of lithium from salt flats. The majority of the world's supply of lithium resides in salt flats around Australia, USA, China and in the Lithium Triangle around Chile, Bolivia and Argentina. Current methods are time-intensive and new techniques are required to satisfy the expected massive increase in demand from electrification. The MOF materials are being developed with University of Texas at Austin and US company Energy Exploration Technologies (EnergyX). |
|
Limitless applications: the 'magic powder' that could prevent future crises
The magic powder (Metal-Organic Frameworks, MOFs) developed by scientists at Monash University and the CSIRO are the potential solutions for the global food and water crisis. One teaspoon of these crystals contain the surface area of an entire football field, and scientists have been able to customise them to absorb and store vast quantities of a desired substance. MOFs can be applied to just about any purpose. The obstacle has long been producing enough of them at a cheap enough cost to be of any practical use. Over in Australia, at the CSIRO, Associate Professor Matt Hill is leading the charge to find a way to make what he calls his “magic powder” commercially viable. In 2008, the team could only produce a teaspoon a week. Today the CSIRO’s startup MOFWORX can produce 10kg per hour on their pilot reactor using a continuous process 20 times more efficient than the conventional batch process. |
|
|
The new faces of science - Dr Matthew Hill in CSIRO Seven group
In 2015, Dr. Matthew Hill was among the CSIRO seven brightest young scientists. CSIRO has pulled seven of its brightest young scientists out of their labs and enlisted them in a campaign to recruit future thinkers, innovators, scientists and entrepreneurs. Known as the CSIRO seven, the group comes from different areas of the organisation’s science and also from across its 54 sites around Australia. |
|
|
Dr. Matthew Hill is the recipient of 2014 Malcolm McIntosh Prize for Physical Scientist of the Year
For his work on the development of metal–organic frameworks for practical industrial application, Dr Matthew Hill, Australian Research Council Future Fellow and leader of the Integrated Nanoporous Materials team at CSIRO, has been awarded the 2014 Malcolm McIntosh Prize for Physical Scientist of the Year. |
|
2014 Australia-Korea Foundation Grant for "Contemporary Materials for Carbon Capture"
In 2014, Dr. Cher Hon (Sam) Lau has been awarded a grant from the Department of Foreign Affairs and Trade (DFAT) to work on “Contemporary Materials for Carbon Capture” with Professor Young-Moo Lee from Hanyang University, Korea. The project focuses on using a new class of porous materials, known as metal organic frameworks to improve the separation properties of plastics. The tunable empty spaces in metal organic frameworks will be utilized for enhanced molecular transport and alternative energy storage of plastics. |
|
"Game changing crystals will clean the air" Dr. Marta Rubio Martinez in Fresh Science National Event
This is a game changer for MOF technology. We increased the production yield from only 1 gram in ten days to several kilograms per hour, without any loss in quality. These crystals have a sponge-like structure, porous structure, which creates an enormous amount of surface area, where up to 80% of the space around the particles is unoccupied/empty. One gram of these crystals has the same surface as an entire football field. For example, a tank filled with our nanocrystals could store up to ten times more carbon dioxide gas within the same volume. The reactions to create these materials are usually slow and yield only a few milligrams of high quality crystals – not enough to provide cost effective real world applications to, for example, energy and transportation industries. But Instead of making the reaction vessel bigger, we pumped the ingredients through a small tube which gives us enough surface to create the crystals, this dramatically reduced the reaction times from days to minutes explains Marta", revealed Marta. |